However, deacetylation is notorious due to the use of plenty of concentrated, corrosive basic solutions ( Chen et al., 2017). It has been reported that deacetylation is commonly inevitable for the valorization of chitin. 
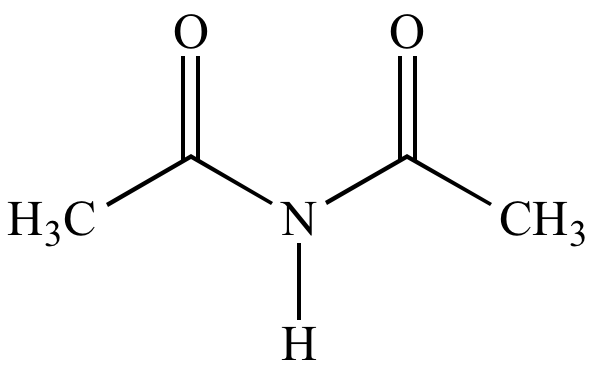
In spite of the enormous economic and environmental interests, the existence of acetyl groups is the main challenge for the exploitation of chitin biomass, which seriously hampers its transformation into fuels and fine chemicals ( Fang and Fan et al., 2019). Chitin is considered as the most promising biomass sources for the production of renewable N-containing chemicals and materials due to its high nitrogen content of around 7% by weight. ( Yabushita et al., 2015 Yan and Chen et al., 2015). Our process provides a strategy that exploiting every functional group adequately in substrates to obtain value-added chemicals.Ĭhitin, the second most abundant biopolymer on the earth after cellulose, consists of N-acetylglucosamine units with ß-1,4-glycosidic linkages. 
Furthermore, the solid residue after removing the acetyl group (denoted as De-chitin) with the sufficient exposure of -NH 2 groups as a solid base catalyst shows excellent performance in the aldol condensation reaction of furfural and acetone to produce fuel precursors. A series of amine derivatives, mainly including aliphatic amine, cyclic amine and functionalized aromatic amine, could be selectively converted into the corresponding amide products frequently found in pharmaceuticals. Herein, for the first time we demonstrate a novel approach to achieve the selective utilization of the N-acetyl group in chitin for transamidation of chitin with amines. The selective conversion of robust chitin commonly requires considerable base catalysts to remove the N-acetyl group as a byproduct in advance, which is non-compliance with the principle of atomic economy. However, the N-acetyl groups in chitin construct strong hydrogen bond networks, which restricts its depolymerization and transformation. The selective transformation of chitin into various renewable N-containing chemicals and medicines has attracted increasing attention. 3Physical Science Laboratory, Huairou National Comprehensive Science Center, Beijing, China.2School of Chemistry and Chemical Engineering, University of Chinese Academy of Sciences, Beijing, China.1Beijing National Laboratory for Molecular Sciences, CAS Key Laboratory of Colloid and Interface and Thermodynamics, CAS Research/Education Center for Excellence in Molecular Sciences, Institute of Chemistry, Chinese Academy of Sciences, Beijing, China.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
